The Epoch Times published an explosive report today stating that they have received emails that allegedly show that the U.S. Food and Drug Administration (FDA) and National Institutes of Health (NIH) have been quietly studying neurological problems that have appeared in people after they took a COVID-19 vaccine.
The report states:
Two U.S. agencies have been quietly studying neurological problems that have appeared in people who have had COVID-19 vaccines, The Epoch Times has found.
The Food and Drug Administration (FDA) and National Institutes of Health (NIH) have been conducting separate research projects into post-vaccination neurological issues, which have manifested with symptoms like facial paralysis and brain fog and have been linked in some cases with the vaccines, according to emails reviewed by The Epoch Times.
One attempt to gain understanding of a problem that experts around the world are struggling to understand is being carried out by Dr. Janet Woodcock, who was acting commissioner of the FDA until Feb. 17.
Woodcock, now the FDA’s principal deputy director, has been personally evaluating neurologic side effects from the COVID-19 vaccines since at least Sept. 13, 2021, according to the emails, many of which have not been reported on previously. FDA epidemiologists are also gathering data to look into the issues, according to messages from Dr. Peter Marks, another top FDA official.
A team at the NIH’s National Institute of Neurological Disorders and Stroke (NINDS), meanwhile, started seeing patients reporting vaccine injuries for a study in early 2021 after receiving complaints shortly after the vaccines were made available. A portion of the patients was examined in person at the Bethesda, Maryland, facility.
None of the reviews or studies appear to have been announced, and health officials have said little about them publicly, despite a growing recognition among experts that at least some issues are likely linked to the vaccines. (Full article here. Copy here with no paywall.)
An examination of the FDA and CDC’s Vaccine Adverse Events Reporting System (VAERS) confirms that deaths and injuries due to neurological issues following COVID-19 injections have skyrocketed, when compared to the same adverse events reported following all FDA-approved vaccines for the previous 30 years.
And while none of the U.S. Health Agencies have published or made public their research on these post-vaccine neurological injuries, many medical journals have.
The COVID-19 experimental vaccines, which were given emergency use authorization illegally, should have been immediately pulled from the market at least a year ago.
Will these criminals in the pharmaceutical industry who pay billions of dollars to get their products approved by the FDA ever face justice for the mass murder they have caused?
Almost 3000 Percent Increase In Neurological Injuries Following COVID-19 Vaccines
According to the Cleveland Clinic, “neuropathy” is:
damage or dysfunction of one or more nerves that typically results in numbness, tingling, muscle weakness and pain in the affected area. Neuropathies frequently start in your hands and feet, but other parts of your body can be affected too.
Neuropathy, often called peripheral neuropathy, indicates a problem within the peripheral nervous system. Your peripheral nervous system is the network of nerves outside your brain and spinal cord. Your brain and spinal cord make up your central nervous system. Think of the two systems working together this way: Your central nervous system is the central station. It is the control center, the hub from which all trains come and go. Your peripheral nervous system are the tracks that connect to the central station. The tracks (the network of nerves) allow the trains (information signals) to travel to and from the central station (your brain and spinal cord).
Neuropathy results when nerve cells, called neurons, are damaged or destroyed. This disrupts the way the neurons communicate with each other and with the brain. Neuropathy can affect one nerve (mononeuropathy) or nerve type, a combination of nerves in a limited area (multifocal neuropathy) or many peripheral nerves throughout the body (polyneuropathy). (Source.)
To determine the number of neurological injuries and deaths occurring after COVID-19 vaccines, and then compare them to the previous 30 years’ historical data following all other FDA approved vaccines, we need to perform several searches in VAERS.
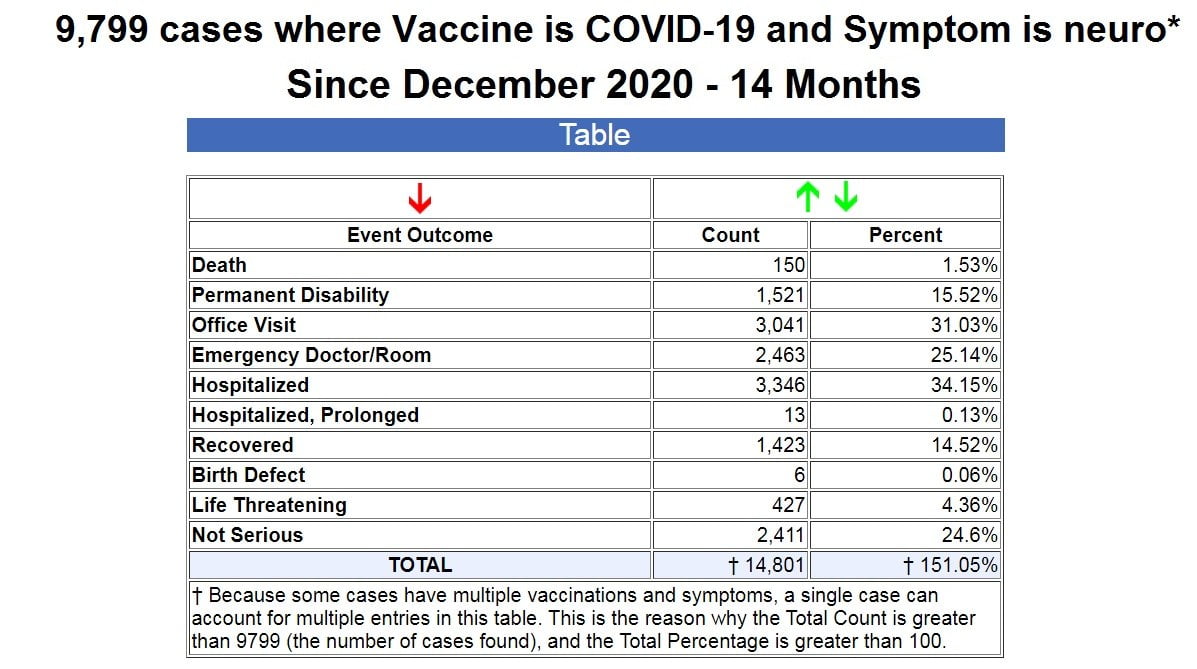
So I began by searching for all symptoms in VAERS that contained “neuro” following COVID-19 shots, and it returned 9,799 cases as of the latest data dump into VAERS this past Friday, which goes through February 11, 2022.
As you can see from the table above, in 14 months since the emergency use authorization of the COVID-19 vaccines, out of the 9,799 reported cases, there have been 150 deaths, 1,521 permanent disabilities, 2,463 ER visits, and 3,359 hospitalizations. (Source.)
By way of comparison, for the previous 30+ years starting in 1990 when VAERS was established, using the exact same search for every symptom containing “neuro,” we get a result of 8,492 cases for ALL FDA-approved vaccines for the past 30+ years combined. (Source.)
The yearly averages then look like this:
- 8399 cases of neurological injuries per year following COVID-19 vaccines
- 283 cases of neurological injuries per year following all FDA-approved vaccines combined
That’s a 2,867 percent increase of neurological injuries following COVID-19 vaccines as compared to all other vaccines approved by the FDA.
Over 3000 Percent Increase In Cases Of Palsy Following COVID-19 Vaccines
There are, of course, other symptoms that are considered neurological conditions that do not contain the word “neuro” in the name of the disease.
Bell’s Palsy, facial paralysis, is one that has been reported frequently following COVID-19 vaccines and is considered a “neurological” injury.
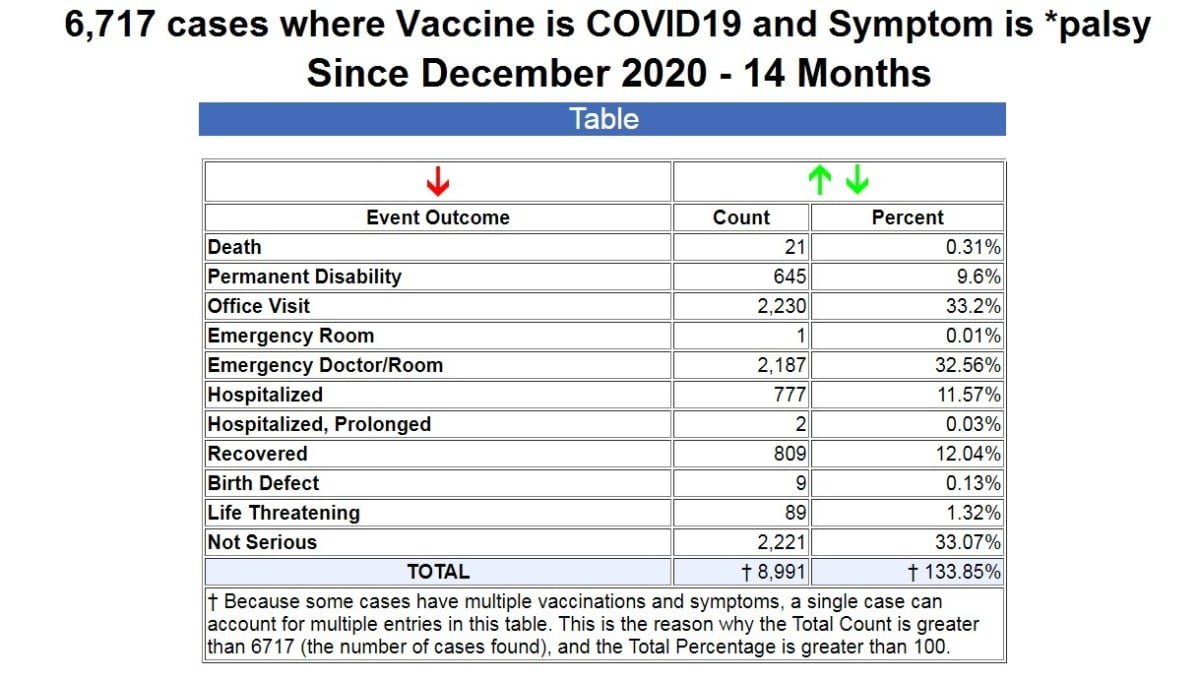
So I searched VAERS for all cases of any kind of “palsy” following COVID-19 vaccines, and it returned a result of 6,717 cases.
As you can see from the table above, in 14 months since the emergency use authorization of the COVID-19 vaccines, out of the 6,717 reported cases, there have been 21 deaths, 645 permanent disabilities, 2,188 ER visits, and 779 hospitalizations. (Source.)
By way of comparison, for the previous 30+ years starting in 1990 when VAERS was established, using the exact same search for every symptom containing “palsy,” we get a result of 4,973 cases for ALL FDA-approved vaccines for the past 30+ years combined. (Source.)
The yearly averages then look like this:
- 5757 cases of “palsy” per year following COVID-19 vaccines
- 166 cases of “palsy” per year following all FDA-approved vaccines combined
That’s a 3,368 percent increase in cases of palsy following COVID-19 vaccines as compared to all other vaccines approved by the FDA.
Over 2000 Percent Increase In Cases Of Sclerosis Following COVID-19 Vaccines
Another neurological injury that has been reported following COVID-19 injections is multiple sclerosis (MS).
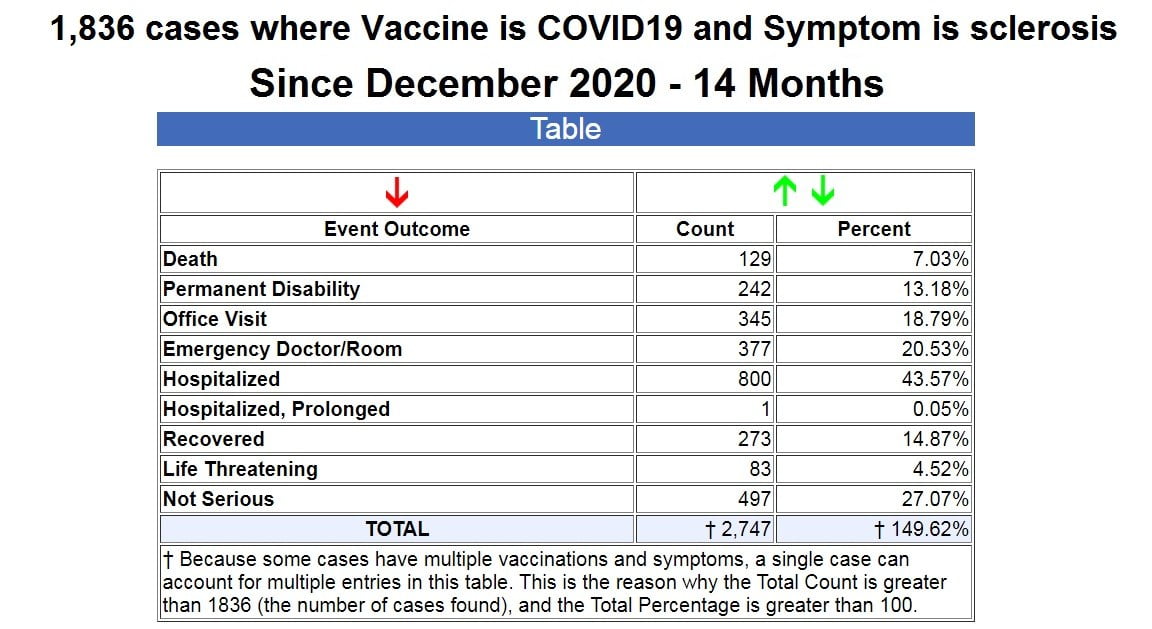
So I searched VAERS for all cases of any kind of “sclerosis” following COVID-19 vaccines, and it returned a result of 1,836 cases.
As you can see from the table above, in 14 months since the emergency use authorization of the COVID-19 vaccines, out of the 1,836 cases of sclerosis, there have been 129 deaths, 242 permanent disabilities, 377 ER visits, and 801 hospitalizations. (Source.)
By way of comparison, for the previous 30+ years starting in 1990 when VAERS was established, using the exact same search for every symptom containing “sclerosis,” we get a result of 1,842 cases for ALL FDA-approved vaccines for the past 30+ years combined. (Source.)
The yearly averages then look like this:
- 1,574 cases of sclerosis per year following COVID-19 vaccines
- 61 cases of sclerosis per year following all FDA-approved vaccines combined
That’s a 2,480 percent increase in cases of sclerosis following COVID-19 vaccines.
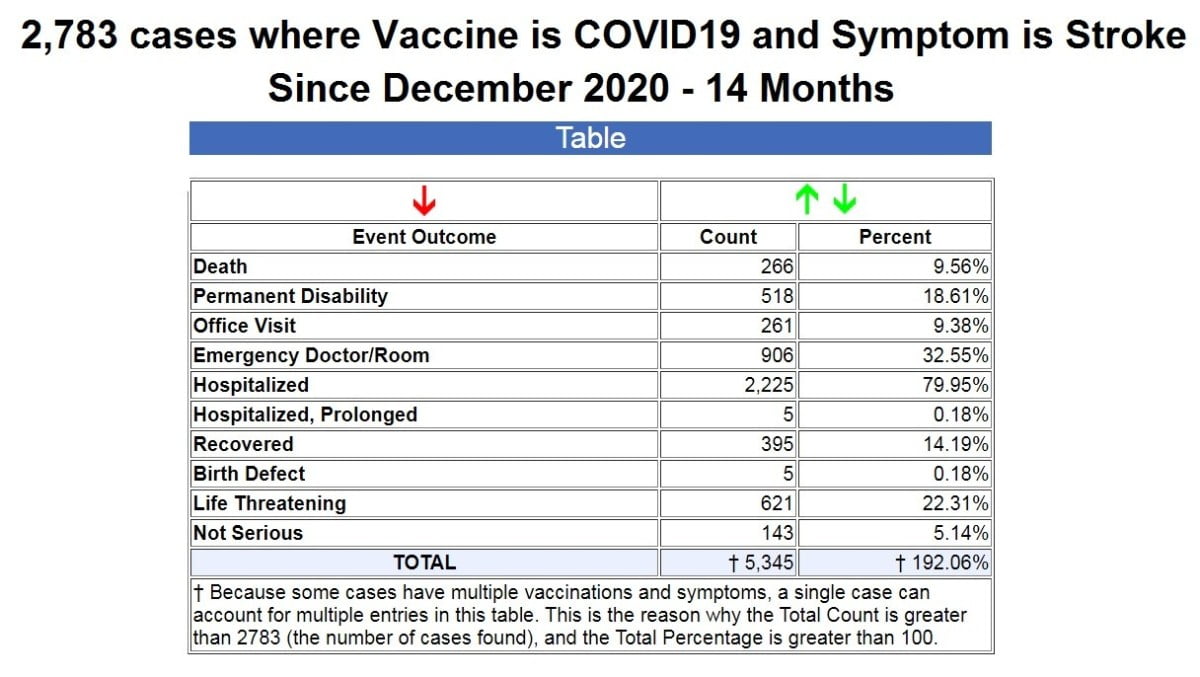
68,000 Percent Increase In Strokes Following COVID-19 Vaccines
Nothing, however, compares to the dramatic increase over reported injuries following COVID-19 vaccines than strokes. While it is probably true that not all strokes can be categorized as “neurological” injuries, the increase is staggering nevertheless.
I searched VAERS for all symptoms containing “stroke” following COVID-19 vaccines, and it returned a result of 2,783 cases. (Source.)
By way of comparison, for the previous 30+ years starting in 1990 when VAERS was established, using the exact same search for every symptom containing “stroke,” we get a result of only 104 cases for ALL FDA-approved vaccines for the past 30+ years combined. (Source.)
The yearly averages then look like this:
- 2385 cases of stroke per year following COVID-19 vaccines
- 3.5 cases of stroke per year following all FDA-approved vaccines combined
That’s an astounding 68,043 percent increase in the cases of stroke following COVID-19 vaccines!
I have run this search and checked my numbers several times, as it just didn’t seem possible that there was that big of an increase in strokes.
So I used a different search term, “ischaemic,” as this is a common medical term associated with strokes, just to see if maybe people started using different terms since the COVID-19 vaccines started that would account for this dramatic increase.
Nope.
Here are the results:
4,575 cases where vaccine is COVID-19 and symptom is “ischaemic.” (Source.)
351 cases for all vaccines for the past 30+ years where symptom is “ischaemic.” (Source.)
The yearly averages:
- 3921 cases per year following COVID-19 vaccines
- 11.7 cases per year following all FDA-approved vaccines combined
That’s also an astounding 33,413 percent increase for ischaemic injuries following COVID-19 vaccines.
What The FDA And NIH Are Not Telling The Public
As one can imagine, the sheer volume of these injuries could not possibly go unnoticed, and thanks to the EPOCH TIMES article today, we know now that the U.S. Government is aware of these numbers, and has been secretly studying them without telling the public.
But here are some others who have published some studies on neurological injuries following COVID-19 vaccines. These are studies published in medical journals and referenced on the U.S. Government’s National Library of Medicine.
- Initial clinical manifestation of multiple sclerosis after immunization with the Pfizer-BioNTech COVID-19 vaccine – Journal of Neuroimmunology
- Multiple cranial nerve palsies following COVID-19 vaccination-Case report – Acta Neurologica Scandinavica
- A rare presentation of undiagnosed multiple sclerosis after the COVID-19 vaccine – Journal of Community Hospital Internal Medical Perspectives
- Case Report: Multiple Sclerosis Relapses After Vaccination Against SARS-CoV2: A Series of Clinical Cases – Frontiers in Neurology
- Immune-Mediated Disease Flares or New-Onset Disease in 27 Subjects Following mRNA/DNA SARS-CoV-2 Vaccination – Journal Vaccines
- Acute bilateral optic/chiasm neuritis with longitudinal extensive transverse myelitis in longstanding stable multiple sclerosis following vector-based vaccination against the SARS-CoV-2 – Journal of Neurology
This is just a sample list, and not exhaustive by any means.
Could it be that the FDA and NIH, which we know are controlled by the pharmaceutical companies, are researching these neurological injuries following COVID-19 shots in order to develop new drugs to treat these diseases, and not to warn the public which would result in fewer people getting the shots?
Here is a report recently written and published at Fierce Pharma that might give a clue to the answer to this question.
Azurity Bags FDA Nod For Grape-Flavored Multiple Sclerosis Drug
The market for novel formulations of baclofen is heating up. Weeks after Saol Therapeutics won approval for its strawberry-flavored oral granules, Azurity Pharmaceuticals has received FDA authorization for an oral suspension aimed at the same piece of the market.
Physicians prescribe baclofen to treat muscle spasms and stiffness in patients with multiple sclerosis and spinal cord injuries. However, the established spinal canal delivery and tablet formulations have certain limitations. Tablets are unsuitable for people who have difficulty swallowing and injections, while getting more of the drug to target sites and addressing some side effects, can be painful.
Over the past two months, two new options have won approval in the U.S. Azurity is the latest entrant to the market, winning FDA approval for a grape-flavored oral suspension formulation of baclofen for use in the treatment of spasticity resulting from multiple sclerosis.
The product, branded Fleqsuvy, is available in 120-ml or 300-ml bottles at a concentration of 5 mg per ml. Azurity is pitching Fleqsuvy as the most concentrated FDA-approved oral liquid baclofen formulation. As Azurity sees things, the concentration of Fleqsuvy is beneficial when treating patients who struggle to swallow as it allows for the lowest volume to be prescribed to patients.
The approval comes two months after FDA authorized Saol’s strawberry-flavored oral baclofen granules. Saol’s granules, which it sells as Lyvispah, are available in 5-mg, 10-mg and 20-mg packs and are approved for administration with or without water, with soft foods and with enteral feeding tubes. (Source.)
The sad thing here is that fruit-flavored medicines are often targeted to children, especially the pediatric age group. With Pfizer’s COVID-19 shot still expected to be approved for infants and toddlers, and children 5 to 11 recently approved, is this a signal for what they expect is coming in the near future?
Source: healthimpactnews.com